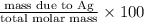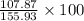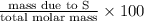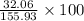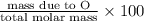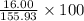Chemistry, 06.01.2020 06:31 heyperdomo8496
1. (nh)2cro-
a) number of moles of h:
b) number of moles of n:
2. ag. so.
a) molar mass:
b) percent composition of ag:
c) percent composition of s:
d) percent composition of o:

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1


You know the right answer?
1. (nh)2cro-
a) number of moles of h:
b) number of moles of n:
2. ag. so.
a) m...
a) number of moles of h:
b) number of moles of n:
2. ag. so.
a) m...
Questions

English, 19.10.2021 20:30

Mathematics, 19.10.2021 20:30

History, 19.10.2021 20:30

Mathematics, 19.10.2021 20:30

Mathematics, 19.10.2021 20:30

English, 19.10.2021 20:30


Social Studies, 19.10.2021 20:30

Geography, 19.10.2021 20:30


World Languages, 19.10.2021 20:30

French, 19.10.2021 20:30


English, 19.10.2021 20:30


Mathematics, 19.10.2021 20:30


Social Studies, 19.10.2021 20:30

Social Studies, 19.10.2021 20:30

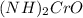 is ammonium Chromate which is monoclinic and yellow Crystal that is formed due to the reaction of ammonium Hydroxide and ammonium di-chromate. It is used as catalyst, corrosion inhibitor as well as analytical inhibitors.
is ammonium Chromate which is monoclinic and yellow Crystal that is formed due to the reaction of ammonium Hydroxide and ammonium di-chromate. It is used as catalyst, corrosion inhibitor as well as analytical inhibitors.