ch4(g)+2o2-> co2(g)+2h2o(g) δh1=-802 kj

Chemistry, 06.01.2020 01:31 nikidastevens36
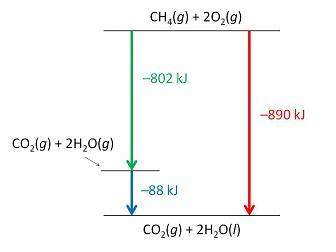
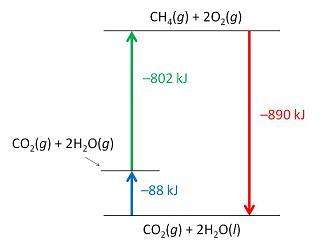
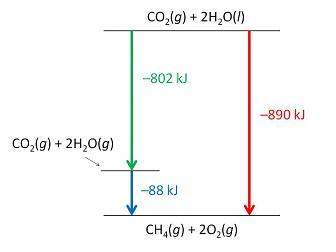
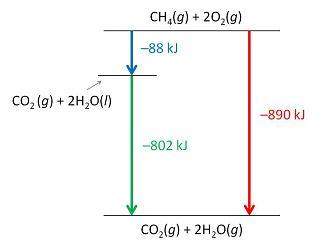
Consider the following intermediate reactions.
ch4(g)+2o2-> co2(g)+2h2o(g) δh1=-802 kj
2h2o(g)-> 2h2o(i) δh2=-88 kj
the overall chemical reaction is as follows.
ch4(g)+2o2(g)-> co2(g)+2h2o(i) δh2=-890 kj
what is the correct enthalpy diagram using the hess law for this system?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:40
Achemistry student weighs out of phosphoric acid , a triprotic acid, into a volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with solution. calculate the volume of solution the student will need to add to reach the final equivalence point. round your answer to significant digits.
Answers: 3

Chemistry, 22.06.2019 05:30
What is the mass defect of a mole of nuclei with 1.8 x 10^15 j/mol binding energy?
Answers: 1

Chemistry, 22.06.2019 05:50
Why doesn't heat added to water make the tempature rise above 100c
Answers: 2

Chemistry, 22.06.2019 22:50
At the current rate, a graph of carbon dioxide produced by fossil fuels over time would slope upward slope downward be horizontal be vertical
Answers: 3
You know the right answer?
Consider the following intermediate reactions.
ch4(g)+2o2-> co2(g)+2h2o(g) δh1=-802 kj
ch4(g)+2o2-> co2(g)+2h2o(g) δh1=-802 kj
Questions

Mathematics, 17.03.2021 23:50

History, 17.03.2021 23:50


Law, 17.03.2021 23:50



English, 17.03.2021 23:50


Mathematics, 17.03.2021 23:50






Mathematics, 17.03.2021 23:50



Health, 17.03.2021 23:50

Mathematics, 17.03.2021 23:50

Mathematics, 17.03.2021 23:50



