
Chemistry, 05.01.2020 01:31 Marcus2935
In the chemical equation zn + 2hcl → zncl2 + h2, the reactants are
a. zinc chloride and hydrogen.
b. zinc and hydrogen carbonate.
c. zinc chlorate and water.
d. zinc and hydrochloric acid.
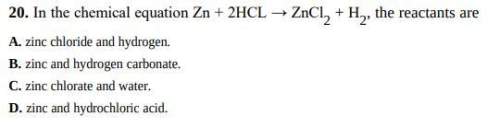

Answers: 3
Another question on Chemistry



Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1

Chemistry, 22.06.2019 23:30
The density of benzene at 15 °c is 0.8787 g/ml. calculate the mass of 0.1500 l of benzene at this temperature. enter your answer in terms of grams
Answers: 2
You know the right answer?
In the chemical equation zn + 2hcl → zncl2 + h2, the reactants are
a. zinc chloride and h...
a. zinc chloride and h...
Questions

English, 08.07.2019 12:00


Mathematics, 08.07.2019 12:00


Mathematics, 08.07.2019 12:00

Mathematics, 08.07.2019 12:00

Mathematics, 08.07.2019 12:00



Spanish, 08.07.2019 12:00



Geography, 08.07.2019 12:00

World Languages, 08.07.2019 12:00

Mathematics, 08.07.2019 12:00


History, 08.07.2019 12:00

Mathematics, 08.07.2019 12:00

Mathematics, 08.07.2019 12:00



