
Astudent is doing a titration using potassium permanganate solution, kmno4, to determine the amount of h2o2 in a sample. the balanced equation for the reaction in the titration is given below:
2 mno4-(aq) + 6 h+(aq) + 5 h2o2(aq) --> ž 2 mn2+(aq) + 8 h2o(l) + 5 o2(g)
a student calculates an amount of moles of h2o2 that is larger than the actual value. which of the following errors could correctly explain the larger value?
the student failed to wear goggles.
the student did not swirl the flask appropriately and therefore stopped short of the endpoint.
the student failed to rinse the buret with kmno4¬ solution after rinsing it with distilled water.
the student added an extra 15 ml of distilled water to the h2o2 solution.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:20
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 03:10
Agas diffuses 1/7 times faster than hydrogen gas (h2). what is the molar mass of the gas? 100.10 g/mol 98.78 g/mol 86.68 g/mol 79.98 g/mol
Answers: 3

Chemistry, 22.06.2019 06:00
How many atoms of mg are present in 97.22 grams of mg? 6.022 × 1023 2.408 × 1024 4.818 × 1024 5.855 × 1025
Answers: 2
You know the right answer?
Astudent is doing a titration using potassium permanganate solution, kmno4, to determine the amount...
Questions

Mathematics, 06.07.2021 14:20


Mathematics, 06.07.2021 14:20




Mathematics, 06.07.2021 14:20

Mathematics, 06.07.2021 14:20

Law, 06.07.2021 14:20

Mathematics, 06.07.2021 14:20

Mathematics, 06.07.2021 14:20

Physics, 06.07.2021 14:20

Mathematics, 06.07.2021 14:20

Social Studies, 06.07.2021 14:20



Computers and Technology, 06.07.2021 14:20

English, 06.07.2021 14:20


Mathematics, 06.07.2021 14:20

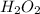 solution.
solution.

