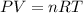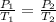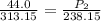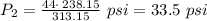Chemistry, 28.12.2019 03:31 brooke0713
The pressure inside an automobile tire is 44.0 pounds per square inch (psi) on a very warm summer day (40.0 °c). calculate the tire pressure in the same tire on a very cold winter day (-35.0 °c) five months later, assuming that no air escapes from the tire. hint: you will need to make another reasonable assumption that is not stated here.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Sulfuric acid (a component of acid rain) reacts with limestone (calcium carbonate) to produce calcium sulfate and carbon dioxide. this damages buildings and statues made of limestone. which solution of sulfuric acid will damage these structures more quickly? a. 0.001% b. 0.005% c. 0.010% d. 0.015%
Answers: 3


Chemistry, 22.06.2019 22:00
Scientists often have to deal with numbers that are either very large or very small. for example, the radius of the sun is approximately 696,000 kilometers, while bacterial cells are as small as 1.9 × 10-4 millimeters. express each number in an alternate form.
Answers: 1

Chemistry, 23.06.2019 11:30
Which of the following is a property of an acid solution? a. slippery to the touch b. ph less than 7 c. turns red litmus paper blue d. bitter taste
Answers: 1
You know the right answer?
The pressure inside an automobile tire is 44.0 pounds per square inch (psi) on a very warm summer da...
Questions






Biology, 24.07.2019 06:00

Biology, 24.07.2019 06:00

History, 24.07.2019 06:00

Chemistry, 24.07.2019 06:00



Social Studies, 24.07.2019 06:00

Social Studies, 24.07.2019 06:00