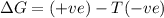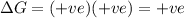Acertain process has δh° > 0, δs° < 0, and δg° > 0. the values of δh° and δs° do not depend on the temperature. which of the following is a correct conclusion about this process? none of the above conclusions is correct. it is non-spontaneous at all t. it is spontaneous at low t. it is spontaneous at all t. it is spontaneous at high t

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:00
Nickel crystallizes in the face-centered cubic (fcc) lattice. the density of the metal is 8902 kg/m3. calculate the radius of a nickel atom.
Answers: 1

Chemistry, 22.06.2019 05:00
Agas can holds 2.0 gal of gasoline. what is this quantity in cubic centimeters?
Answers: 2

Chemistry, 22.06.2019 06:00
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2

Chemistry, 22.06.2019 15:30
A1.5l container holds p.50 grams of an unknown gas at a pressure of 0.44 atm and a temperature of 50.c what is the molar mass of the unknown gas
Answers: 1
You know the right answer?
Acertain process has δh° > 0, δs° < 0, and δg° > 0. the values of δh° and δs° do not dep...
Questions

Mathematics, 30.10.2020 17:40

Mathematics, 30.10.2020 17:40





Mathematics, 30.10.2020 17:40


Mathematics, 30.10.2020 17:40

Mathematics, 30.10.2020 17:40


Computers and Technology, 30.10.2020 17:40


Mathematics, 30.10.2020 17:40



Mathematics, 30.10.2020 17:40

Mathematics, 30.10.2020 17:40


Biology, 30.10.2020 17:40

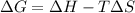
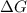 = Gibbs free energy = +ve
= Gibbs free energy = +ve
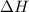 = enthalpy change = +ve
= enthalpy change = +ve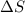 = entropy change = -ve
= entropy change = -ve