
Chemistry, 27.12.2019 02:31 camillesmith6630
Calculate δs° for the reaction: 4cr(s) + 3o2(g) → 2cr2o3(s), substance: cr(s) o2(g) cr2o3(s), s°(j/k⋅mol): 23.77 205.138 81.2

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 07:20
The diagrams show objects’ gravitational pull toward each other. which statement describes the relationship between diagram x and y? gravity attracts only larger objects toward one another. gravity attracts larger objects only if they are close to one another. if the masses of the objects increase, then the force between them also increases. if distance between the objects increases, then the amount of force also increases.
Answers: 1

Chemistry, 22.06.2019 19:00
How many liters of ethylene glycol antifreeze (c2h6o2), with a density of 1.100 g/l, would you add to your car radiator containing 15.0 kg of water if you needed to protect your engine to - 21.5°c? for water, kf = 1.86°c m -1.
Answers: 1

You know the right answer?
Calculate δs° for the reaction: 4cr(s) + 3o2(g) → 2cr2o3(s), substance: cr(s) o2(g) cr2o3(s), s°(j...
Questions




Mathematics, 28.08.2019 23:50

Mathematics, 28.08.2019 23:50



Mathematics, 28.08.2019 23:50

Chemistry, 28.08.2019 23:50

Mathematics, 28.08.2019 23:50


English, 28.08.2019 23:50

Social Studies, 28.08.2019 23:50

Social Studies, 28.08.2019 23:50

Mathematics, 28.08.2019 23:50

English, 28.08.2019 23:50


Biology, 28.08.2019 23:50

History, 28.08.2019 23:50

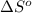 for the reaction is 1051.93 J/K
for the reaction is 1051.93 J/K![\Delta S^o_{rxn}=\sum [n\times \Delta S^o_{(product)}]-\sum [n\times \Delta S^o_{(reactant)}]](/tpl/images/0434/2862/52737.png)
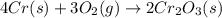
![\Delta S^o_{rxn}=[(2\times \Delta S^o_{(Cr_2O_3(s))})]-[(4\times \Delta S^o_{(Cr(s))})+(3\times \Delta S^o_{(O_2(g))})]](/tpl/images/0434/2862/e2e78.png)
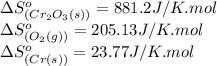
![\Delta S^o_{rxn}=[(2\times (881.2))]-[(4\times (23.77))+(3\times (205.13))]\\\\\Delta S^o_{rxn}=1051.93J/K](/tpl/images/0434/2862/00c14.png)


