
Chemistry, 27.12.2019 00:31 ehuntsman8221
Asolution is prepared by dissolving 0.56 g of benzoic acid (c6h5co2h, ka 6.4 ) in enough water to make 1.0 l of solution. calculate [c6h5co2h], , , , and the ph of this solution.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:00
From the options provided for each element below, choose the properties that it may have based on its location in the periodic table fluorine (f): highly reactive nonmetal shiny a conductor
Answers: 1

Chemistry, 23.06.2019 09:00
Which factor is likely to impact the possible number of compounds? presence of unlimited number of elements in the periodic table the inability of atoms to align perfectly with other atoms the ability of all elements to react with every other element all elements being equally reactive
Answers: 2

Chemistry, 24.06.2019 03:30
Which of these elements exist as a liquid at normal earth temperatures?
Answers: 2

Chemistry, 24.06.2019 08:00
Asolution has a ph of 4.20. using the relationship between ph and poh, what is the concentration of oh−? a. 9.9 × 10-1 m b. 6.2 × 10-1 m c. 6.3 × 10-5 m d. 6.7 × 10-6 m e. 1.6 × 10-10 m
Answers: 3
You know the right answer?
Asolution is prepared by dissolving 0.56 g of benzoic acid (c6h5co2h, ka 6.4 ) in enough water to...
Questions

Mathematics, 25.08.2019 04:10


English, 25.08.2019 04:10


History, 25.08.2019 04:10

Mathematics, 25.08.2019 04:10

Social Studies, 25.08.2019 04:10


Computers and Technology, 25.08.2019 04:10



Mathematics, 25.08.2019 04:10

Mathematics, 25.08.2019 04:10


Mathematics, 25.08.2019 04:10





Mathematics, 25.08.2019 04:10

![$\left[\mathrm{H}^{+}\right]=\left[\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CO}_{2}^{-}\right]=5.1 \times 10^{-4} \mathrm{M}$](/tpl/images/0434/1977/10f45.png)
![$\left[\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CO}_{2} \mathrm{H}\right]=4.1 \times 10^{-3} \mathrm{M}$](/tpl/images/0434/1977/9a3e2.png)
![$\left[\mathrm{OH}^{-}\right]=1.9 \times 10^{-11} \mathrm{M}](/tpl/images/0434/1977/e5b4e.png)
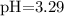
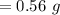
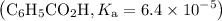
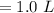
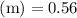
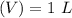
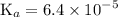
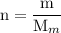
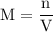
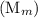 and the number of moles (n) of benzoic acid:
and the number of moles (n) of benzoic acid: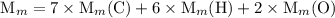
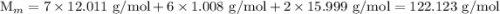
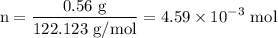
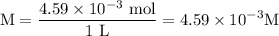
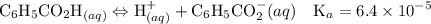
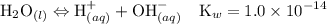
![$\mathrm{K}_{a}=\frac{\left[\mathrm{H}^{+}\right]\left[\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CO}_{2}^{-}\right]}{\left[\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CO}_{2} \mathrm{H}\right]}=6.4 \times 10^{-5}$](/tpl/images/0434/1977/e74c4.png)
![$\left[\mathrm{H}^{+}\right]_{0}$](/tpl/images/0434/1977/f38bd.png) is 0. (because of autonization)
is 0. (because of autonization)![$\left[\mathrm{H}^{+}\right]_{0}=10^{-7} \mathrm{M} \approx 0$](/tpl/images/0434/1977/a3a3d.png)
![$\left[\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CO}_{2} \mathrm{H}\right]=\left[\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CO}_{2} \mathrm{H}\right]_{0}-\mathrm{x}=0.00459-\mathrm{x}$](/tpl/images/0434/1977/a086a.png)
![$\left[\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CO}_{2}^{-}\right]=\left[\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CO}_{2}^{-}\right]_{0}+\mathrm{x}=0+\mathrm{x}=\mathrm{x}$](/tpl/images/0434/1977/89a6e.png)
![$\left[\mathrm{H}^{+}\right]=\left[\mathrm{H}^{+}\right]_{0}+x=0+x=x$](/tpl/images/0434/1977/2b64c.png)
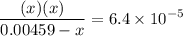
![$\left[\mathrm{H}^{+}\right]$](/tpl/images/0434/1977/93e23.png) is much smaller than
is much smaller than 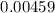 to decide whether the following approximation is valid or not:
to decide whether the following approximation is valid or not: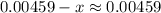
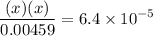
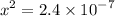
![$\mathrm{x}=4.9 \times 10^{-4}=\left[\mathrm{H}^{+}\right]$\\](/tpl/images/0434/1977/329c6.png)
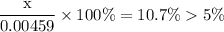
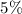 of acid is dissociated.
of acid is dissociated.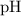 :
: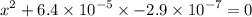
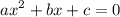
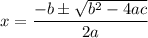
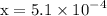
![\mathrm{M}=\left[\mathrm{H}^{+}\right]=\left[\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CO}_{2}^{-}\right]$](/tpl/images/0434/1977/6835a.png)
![$\left[\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CO}_{2} \mathrm{H}\right]=0.00459-0.00051=4.1 \times 10^{-3} \mathrm{M}$](/tpl/images/0434/1977/3ea5f.png)
![$\left[\mathrm{OH}^{-}\right]=\frac{\mathrm{K}_{w}}{\left[\mathrm{H}^{+}\right]}=\frac{1.0 \times 10^{-14}}{5.1 \times 10^{-4}}=1.9 \times 10^{-11} \mathrm{M}$](/tpl/images/0434/1977/7340a.png)
![$\mathrm{pH}=-\log \left[\mathrm{H}^{+}\right]=-\log \left(5.1 \times 10^{-4}\right)=3.29$](/tpl/images/0434/1977/3ad18.png)


