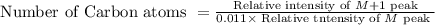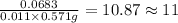Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:40
Determine the energy released per kilogram of fuel used. given mev per reaction, calculate energy in joules per kilogram of reactants. consider 1 mole of tritium plus 1 mole of deuterium to be a mole of “reactions” (total molar mass = 5 grams).
Answers: 1

Chemistry, 22.06.2019 10:50
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1
You know the right answer?
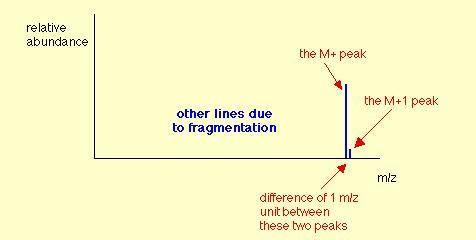
The mass spectrum of an unknown compound has a molecular ion peak with a relative intensity of 57.10...
Questions


English, 07.11.2020 17:40

Social Studies, 07.11.2020 17:40

History, 07.11.2020 17:40



Arts, 07.11.2020 17:40

Mathematics, 07.11.2020 17:40


English, 07.11.2020 17:40


Mathematics, 07.11.2020 17:50


English, 07.11.2020 17:50

English, 07.11.2020 17:50


History, 07.11.2020 17:50

World Languages, 07.11.2020 17:50


Mathematics, 07.11.2020 17:50