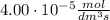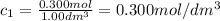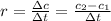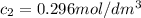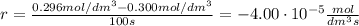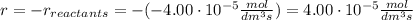Chemistry, 24.12.2019 18:31 hesterkl1225
At the start of a reaction, a 1.00 dm solution contains 0.300 mol of ethanol.
after 100 seconds the concentration of the ethanol has decreased to 0.296 mol/dmº
what is the rate of reaction over the first 100 seconds?
a 2.96 x 10-3 mol/dm/s
b 3.00 x 10 mol/dm/s
c 4.00 x 10 mol/dm®/s
d 8.00 x 10 mol/dm/s

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 04:30
How do i complete this electrolysis of water lab? i’m at home, so i don’t have the materials, and the lab didn’t properly work and was incomplete at school.
Answers: 1

Chemistry, 22.06.2019 15:50
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate
Answers: 1

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3
You know the right answer?
At the start of a reaction, a 1.00 dm solution contains 0.300 mol of ethanol.
after 100 seconds...
after 100 seconds...
Questions

Mathematics, 11.03.2021 17:20

Mathematics, 11.03.2021 17:20

Mathematics, 11.03.2021 17:20

Mathematics, 11.03.2021 17:20

Mathematics, 11.03.2021 17:20

Chemistry, 11.03.2021 17:20


History, 11.03.2021 17:20



Mathematics, 11.03.2021 17:20

Chemistry, 11.03.2021 17:20


Chemistry, 11.03.2021 17:20

Social Studies, 11.03.2021 17:20



Mathematics, 11.03.2021 17:20