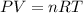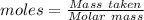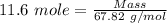Chemistry, 20.12.2019 18:31 hannabeth91
Boron trifluoride gas is collected at 2.0 degree c in an evacuated flask with a measured volume of 15.0 l. when all the gas has been collected, the pressure in the flask is measured to be 0.130 atm. calculate the mass and number of moles of boron trifluoride gas that were collected. be sure your answer has the correct number of significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 20:00
The volume of a single vanadium atom is 9.29×10-24 cm3. what is the volume of a vanadium atom in microliters?
Answers: 3

Chemistry, 23.06.2019 00:10
Apropane torch is lit inside a hot air balloon during preflight preparations to inflate the balloon. which condition of the gas remains constant
Answers: 2

Chemistry, 23.06.2019 03:30
Select the correct lewis structure for fluorine which is group 7a element?
Answers: 1

Chemistry, 23.06.2019 04:10
What does the field of thermodynamics relate to a-changes in nuclear reactions b- changes in energy in systems c changes in molecular structure d changes in atomic properties
Answers: 1
You know the right answer?
Boron trifluoride gas is collected at 2.0 degree c in an evacuated flask with a measured volume of 1...
Questions


Mathematics, 22.02.2021 01:00

Spanish, 22.02.2021 01:00

Biology, 22.02.2021 01:00

Mathematics, 22.02.2021 01:00

Physics, 22.02.2021 01:00

Mathematics, 22.02.2021 01:00

Mathematics, 22.02.2021 01:00

Spanish, 22.02.2021 01:00


Mathematics, 22.02.2021 01:00

Arts, 22.02.2021 01:00

Mathematics, 22.02.2021 01:00

Physics, 22.02.2021 01:00