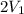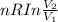Chemistry, 20.12.2019 03:31 arifkarimi9214
Calculate the change in the entropy of the system and also the change in the entropy of the surroundings, and the resulting total change in entropy, when a sample of nitrogen gas of mass 14 g at 298 k and 1.00 bar doubles its volume in (a) an isothermal reversible expansion, (b) an isothermal irreversible expansion against pext = 0, and (c) an adiabatic reversible expansion.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
Pbco3 –> pbo+ co2. how many liters of carbon dioxide gas is produced from the decomposition of 32 grams of lead (ll) carbonate?
Answers: 1

Chemistry, 22.06.2019 03:30
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1

Chemistry, 22.06.2019 06:00
Oxidation-reduction reactions (often called "redox" for short) are reactions that involve the transfer of electrons from one species to another. oxidation states, or oxidation numbers, allow chemists to keep track of these electron transfers. in general, one element will lose electrons (oxidation), with the result that it will increase in oxidation number, and another element will gain electrons (reduction), thereby decreasing in oxidation number. the species that is oxidized is called the reducing agent or reductant. the species that is reduced is called the oxidizing agent or oxidant. to sum up: oxidation = increase in oxidation state = loss of electrons = reducing agent reduction = decrease in oxidation state = gain of electrons = oxidizing agent part a which element is oxidized in this reaction? fe2o3+3co→2fe+3co2 enter the elemental symbol. view available hint(s) is oxidized part b which element is reduced in this reaction? 2hcl+2kmno4+3h2c2o4→6co2+2mno2+2kcl+4h2o enter the elemental symbol. view available hint(s) is reduced
Answers: 1

Chemistry, 22.06.2019 09:30
One way that radioactive waste is treated is by burying it in repositories. the repositories are found only in states with very low populations. true or false? a. trueb. false(also i meant to put high school but it put down middle school instead)
Answers: 1
You know the right answer?
Calculate the change in the entropy of the system and also the change in the entropy of the surround...
Questions




History, 03.04.2020 02:12


World Languages, 03.04.2020 02:12


Social Studies, 03.04.2020 02:12


English, 03.04.2020 02:12

History, 03.04.2020 02:12







Mathematics, 03.04.2020 02:12

Chemistry, 03.04.2020 02:12

History, 03.04.2020 02:12

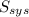 = 2.881 J/K; Δ
= 2.881 J/K; Δ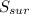 = -2.881 J/K; total change in entropy = 0
= -2.881 J/K; total change in entropy = 0 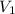
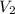 =
=