Chemistry, 20.12.2019 02:31 caydennnnn5764
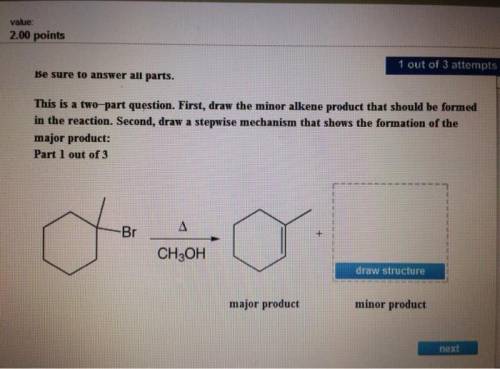
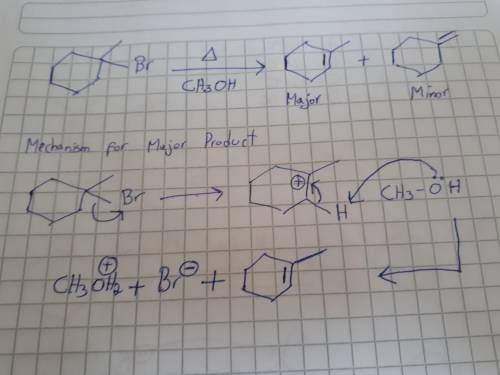
Be sure to answer all parts. this is a two-part question. first, draw the minor alkene product that should be formed in the reaction. second, draw a stepwise mechanism that shows the formation of the major product: part 1: 2xsafari + view structure major product minor product part 2: view structure h5mech30504 view structure + br+ br− ch3obr+ ch3oh2+ part 3 out of 3 edit structure arr edit structure + br+ ch3oh2+ ch3obr+ br−

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 03:00
Compare the valence electron configuration of the nobles gas elements seen here. what statement is correct?
Answers: 2

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 12:30
Sodium sulfate dissolves as follows: na2so4(s) → 2na+(aq) + so42- (aq). how many moles of na2so4 are required to make 1.0 l of solution in which the na concentration is 0.10 m?
Answers: 2
You know the right answer?
Be sure to answer all parts. this is a two-part question. first, draw the minor alkene product that...
Questions





Mathematics, 11.10.2019 23:10

Physics, 11.10.2019 23:10

Mathematics, 11.10.2019 23:10




Chemistry, 11.10.2019 23:10

History, 11.10.2019 23:10

Mathematics, 11.10.2019 23:10



Business, 11.10.2019 23:10



Computers and Technology, 11.10.2019 23:10