

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 22.06.2019 06:00
Calculate the mass of silver needed to react with chlorine to produce 126g if silver chloride?
Answers: 3

Chemistry, 22.06.2019 09:40
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2

Chemistry, 22.06.2019 10:20
In a reaction equation, where are the products located? a.) above the arrow b.) to the right of the arrow c.) to the left of the arrow d.) below the arrow
Answers: 2
You know the right answer?
Which of the following aqueous solutions will have the lowest vapor pressure at 25 degrees celsius?...
Questions


Arts, 10.05.2021 14:00

Mathematics, 10.05.2021 14:00

English, 10.05.2021 14:00

Mathematics, 10.05.2021 14:00


Mathematics, 10.05.2021 14:00

World Languages, 10.05.2021 14:00

Mathematics, 10.05.2021 14:00


Business, 10.05.2021 14:00


English, 10.05.2021 14:00

Biology, 10.05.2021 14:00

English, 10.05.2021 14:00

Social Studies, 10.05.2021 14:00

Social Studies, 10.05.2021 14:00

Chemistry, 10.05.2021 14:00

Mathematics, 10.05.2021 14:00

World Languages, 10.05.2021 14:00

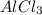
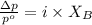
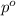 = vapor pressure of the pure component (water)
= vapor pressure of the pure component (water)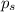 = vapor pressure of the solution
= vapor pressure of the solution 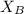 = mole fraction of solute
= mole fraction of solute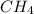 is not possible because it is a non-electrolyte solute. So, the Van't Hoff factor will be, 1.
is not possible because it is a non-electrolyte solute. So, the Van't Hoff factor will be, 1.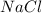 will be,
will be,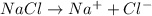
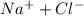 = 1 + 1 = 2
= 1 + 1 = 2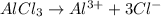
 = 1 + 3 = 4
= 1 + 3 = 4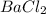 will be,
will be,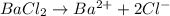
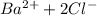 = 1 + 2 = 3
= 1 + 2 = 3

