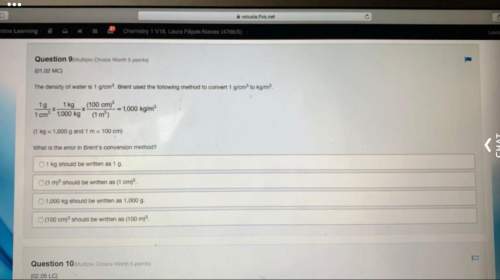
When baking soda (sodium bicarbonate or sodium hydrogen carbonate, nahco3) is heated, it releases carbon dioxide gas, which is responsible for the rising of cookies, doughnuts, and bread.
a) write a balanced equation for the decomposition of the compound (one of the products is na2co3).
b) calculate the mass of nahco3 required to produce 20.5 g of co2.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 10:10
Stage in which a star’s outer layers have started to cool and grow outward?
Answers: 3

Chemistry, 22.06.2019 20:30
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2

Chemistry, 23.06.2019 11:00
Achemist weighed out 101.g of silver. calculate the number of moles of silver she weighed out.
Answers: 2
You know the right answer?
When baking soda (sodium bicarbonate or sodium hydrogen carbonate, nahco3) is heated, it releases ca...
Questions


Computers and Technology, 12.02.2021 18:30


Mathematics, 12.02.2021 18:30

Mathematics, 12.02.2021 18:30

Mathematics, 12.02.2021 18:30



Mathematics, 12.02.2021 18:30


Mathematics, 12.02.2021 18:30

Mathematics, 12.02.2021 18:30

English, 12.02.2021 18:30



History, 12.02.2021 18:30

Chemistry, 12.02.2021 18:30

Arts, 12.02.2021 18:30

Mathematics, 12.02.2021 18:30

History, 12.02.2021 18:30