
Chemistry, 18.12.2019 19:31 cyaransteenberg
At a given temperature, the elementary reaction a − ⇀ ↽ − b , in the forward direction, is first order in a with a rate constant of 0.0340 s − 1 . the reverse reaction is first order in b and the rate constant is 0.0740 s − 1 . what is the value of the equilibrium constant for the reaction a − ⇀ ↽ − b at this temperature?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3

Chemistry, 22.06.2019 14:20
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1

Chemistry, 23.06.2019 00:50
What is the enthalpy of combustion (per mole) of c4h10 (g)? –2,657.5 kj/mol –5315.0 kj/mol –509.7 kj/mol –254.8 kj/mol
Answers: 1
You know the right answer?
At a given temperature, the elementary reaction a − ⇀ ↽ − b , in the forward direction, is first ord...
Questions


Chemistry, 19.10.2019 04:00

History, 19.10.2019 04:00



History, 19.10.2019 04:00


Mathematics, 19.10.2019 04:00

Mathematics, 19.10.2019 04:00

Mathematics, 19.10.2019 04:00

History, 19.10.2019 04:00


Social Studies, 19.10.2019 04:00



Health, 19.10.2019 04:00

Mathematics, 19.10.2019 04:00


Mathematics, 19.10.2019 04:00

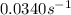
![R=K[A]](/tpl/images/0424/5632/3dc0d.png)
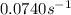
![R'=K'[B]](/tpl/images/0424/5632/2c128.png)
![K[A]=K'[B]](/tpl/images/0424/5632/af2e7.png)
![K_{eq}=\frac{[B]}{[A]}=\frac{K}{K'}=\frac{0.0340 s^{-1}}{0.0740 s^{-1}}=0.4595](/tpl/images/0424/5632/574c7.png)


