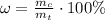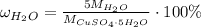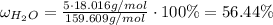Chemistry, 18.12.2019 05:31 jesussaves333
For hydrated copper (ii) sulfate calculate the mass percentage of water in your hydrated crystals.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3



Chemistry, 23.06.2019 01:00
Animals that reproduce sexually either do it through external or internal fertilization. read the following statement and decide if it is true or false. birds reproduce through external reproduction which is because the female will then be able to protect the egg.
Answers: 1
You know the right answer?
For hydrated copper (ii) sulfate calculate the mass percentage of water in your hydrated crystals....
Questions

Spanish, 06.05.2021 02:00

Mathematics, 06.05.2021 02:00




Mathematics, 06.05.2021 02:00



Social Studies, 06.05.2021 02:00

Mathematics, 06.05.2021 02:00


History, 06.05.2021 02:00


Mathematics, 06.05.2021 02:00

Mathematics, 06.05.2021 02:00

Mathematics, 06.05.2021 02:00

German, 06.05.2021 02:00



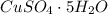 .
.