Chemistry, 18.12.2019 04:31 angelvega2003
The great french chemist antoine lavoisier discovered the law of conservation of mass in part by doing a famous experiment in 1775. in this experiment lavoisier found that mercury(ii) oxide, when heated, decomposed into liquid mercury and an invisible and previously unknown substance: oxygen gas.
1. write a balanced chemical equation, including physical state symbols, for the decomposition of solid mercury(ii) oxide (hgo) into liquid mercury and gaseous dioxygen.
2. suppose 50.0ml of dioxygen gas are produced by this reaction, at a temperature of 90°c and pressure of exactly 1atm. calculate the mass of mercury(ii) oxide that must have reacted. be sure your answer has the correct number of significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 13:00
Is 9 correct? and can someone me with 10? it’s due tomorrow, you
Answers: 1

Chemistry, 22.06.2019 14:00
Displacement is the slope of a velocity vs. time graph a. true b. false
Answers: 1

Chemistry, 22.06.2019 16:00
How do dying stars contribute to the formation of planets
Answers: 1

Chemistry, 22.06.2019 18:00
The fact that the total amount of energy in a system remains constant is a(n)
Answers: 1
You know the right answer?
The great french chemist antoine lavoisier discovered the law of conservation of mass in part by doi...
Questions






Spanish, 26.06.2020 19:01


Mathematics, 26.06.2020 19:01



English, 26.06.2020 19:01


Mathematics, 26.06.2020 19:01


Mathematics, 26.06.2020 19:01

Mathematics, 26.06.2020 19:01



English, 26.06.2020 19:01

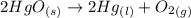
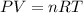
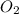 = 0.001677 mol
= 0.001677 mol moles of mercury(II) oxide are reacted
moles of mercury(II) oxide are reacted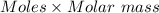 =
=