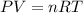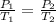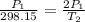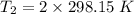Gsuppose a sample of an ideal gas in a container is subjected to a temperature change. a decrease in temperature will the kinetic energy and average speed of the gas particles. as a result, the pressure on the walls of the container will if the gas starts at 25 ∘ c, what temperature would the gas need to reach for its pressure to double? temperature = ∘ c

Answers: 3
Another question on Chemistry



Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

You know the right answer?
Gsuppose a sample of an ideal gas in a container is subjected to a temperature change. a decrease in...
Questions

Computers and Technology, 24.12.2021 05:50




SAT, 24.12.2021 06:00

Computers and Technology, 24.12.2021 06:00


English, 24.12.2021 06:00

Chemistry, 24.12.2021 06:00

SAT, 24.12.2021 06:00


Social Studies, 24.12.2021 06:00




SAT, 24.12.2021 06:00



Health, 24.12.2021 06:00

History, 24.12.2021 06:00