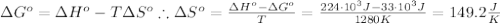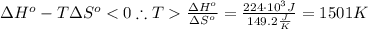The standard reaction enthalpy of zn(s) + h2o(g) →zno(s) + h2(g) is known to be hr 0 = 224 kj and is approximately constant from 920 k up to 1280 k. the standard reaction free energy is +33 kj at 1280 k. calculate the equilibrium constant at 1280 k and then calculate the temperature at which the equilibrium constant becomes greater than 1.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:00
The earth's moon is unusually large. two popular theories of the moon's origin include the "sister world" hypothesis, which states that the moon formed from the same materials as the earth, near enough to the earth that they fell into orbit around each other. a second theory is the "capture" hypothesis, in which the moon formed elsewhere in the solar system, and the earth's gravity pulled it into its orbit. studies of what the moon is made of indicate that some of its materials had to come from the earth or from the same area of the solar system where the earth had formed. at the same time, the moon does not contain much of the material that makes up the earth's core, so the moon could not have formed from the same materials as the earth. how do the two facts above affect the described theories of the moon's origin? a. they show that scientists will never agree on where the moon came from. b. they show that more experiments on moon formation need to be done. c. they show that no theory accounts for the existence of the moon. d. they show that neither theory is complete and entirely correct.
Answers: 2

Chemistry, 22.06.2019 08:00
An observation that requires measurement is called quantitative observable or qualitative
Answers: 1

Chemistry, 22.06.2019 14:30
What type(s) of intermolecular forces are expected between ch3ch2cooh molecules? dipole forces, induced dipole forces, hydrogen bonding
Answers: 1

Chemistry, 22.06.2019 20:30
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2
You know the right answer?
The standard reaction enthalpy of zn(s) + h2o(g) →zno(s) + h2(g) is known to be hr 0 = 224 kj and i...
Questions

History, 03.12.2021 07:20



Chemistry, 03.12.2021 07:20












Mathematics, 03.12.2021 07:20




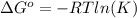
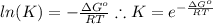
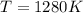 and the ideal gas law constant
and the ideal gas law constant 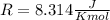 , we obtain:
, we obtain: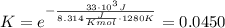
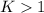 , then
, then 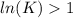 and
and 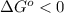 .
.