
Chemistry, 17.12.2019 23:31 cathydaves
Carbon monoxide reacts with oxygen gas to form carbon dioxide according to the following chemical equation. 2co(g)+o2(g)⟶2co2(g) how many ml of oxygen gas will remain in the reaction vessel when 82.4ml of carbon monoxide gas reacts with 80.0ml of oxygen gas to form carbon dioxide under constant pressure and temperature?

Answers: 1
Another question on Chemistry


Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

You know the right answer?
Carbon monoxide reacts with oxygen gas to form carbon dioxide according to the following chemical eq...
Questions

Mathematics, 30.06.2019 14:30

Health, 30.06.2019 14:30

History, 30.06.2019 14:30

English, 30.06.2019 14:30

Mathematics, 30.06.2019 14:30

Mathematics, 30.06.2019 14:30


Geography, 30.06.2019 14:30

Mathematics, 30.06.2019 14:30

English, 30.06.2019 14:30

Advanced Placement (AP), 30.06.2019 14:30

Mathematics, 30.06.2019 14:30


Biology, 30.06.2019 14:30

Physics, 30.06.2019 14:30



Physics, 30.06.2019 14:30

Mathematics, 30.06.2019 14:30

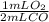 = 41.2 mL O₂
= 41.2 mL O₂


