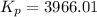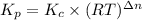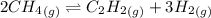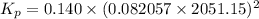Chemistry, 17.12.2019 00:31 jordantay208
For chemical reactions involving ideal gases, the equilibrium constant k can be expressed either in terms of the concentrations of the gases (in m) or as a function of the partial pressures of the gases (in atmospheres). in the latter case, the equilibrium constant is denoted as kp to distinguish it from the concentration-based equilibrium constant kc (sometimes referenced as just k).for the reaction 2ch4(g)⇌c2h2(g)+3h2(g) kc = 0.140 at 1778 ∘c . what is kp for the reaction at this temperature? express your answer numerically.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:00
In the cathode ray tube experiment, j. j. thomson passed an electric current through different gases inside a cathode ray tube in the presence of an electric field. in which two ways did this experiment change scientists’ understanding of the atom?
Answers: 2


Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1

Chemistry, 22.06.2019 22:30
Rank the four gases (air, exhaled air, gas produced from from decomposition of h2o2, gas from decomposition of nahco3) in order of increasing concentration of co2
Answers: 1
You know the right answer?
For chemical reactions involving ideal gases, the equilibrium constant k can be expressed either in...
Questions

Mathematics, 14.04.2021 01:30

Mathematics, 14.04.2021 01:30


History, 14.04.2021 01:30

Health, 14.04.2021 01:30

Spanish, 14.04.2021 01:30

Mathematics, 14.04.2021 01:30

Mathematics, 14.04.2021 01:30


Mathematics, 14.04.2021 01:30


English, 14.04.2021 01:30

Chemistry, 14.04.2021 01:30




Mathematics, 14.04.2021 01:30



Social Studies, 14.04.2021 01:30