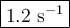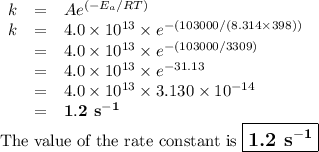Chemistry, 16.12.2019 22:31 caveman171
If the activation energy for a given compound is found to be 103 kj/mol, with a frequency factor of 4.0 × 1013 s-1, what is the rate constant for this reaction at 398 k? 2.5 × 107 s-18.2 s-13.9 × 1010 s-11.2 s-11.7 × 1010 s-1

Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 05:30
Why is soap used to remove grease? a. its nonpolar end dissolves the grease. b. it makes the water bond with the grease. c. it chemically bonds with the grease. d. its polar end dissolves the grease.correct answer for apex - a, its nonpolar end dissolves the grease.
Answers: 1

Chemistry, 22.06.2019 07:30
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3
You know the right answer?
If the activation energy for a given compound is found to be 103 kj/mol, with a frequency factor of...
Questions







Biology, 05.05.2020 07:42


Chemistry, 05.05.2020 07:42


History, 05.05.2020 07:42

Mathematics, 05.05.2020 07:42

Mathematics, 05.05.2020 07:42


History, 05.05.2020 07:42




Mathematics, 05.05.2020 07:42