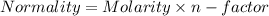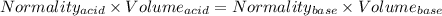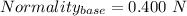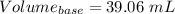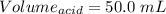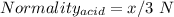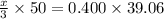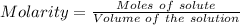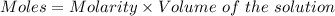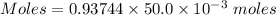Chemistry, 16.12.2019 19:31 secondcolinmills005
You are given a 1.00 g sample of an unknown tri-protic acid, which you dissolve in 50.0 ml of water containing phenolphthalein indicator. you titrate the acid solution with standardized 0.400 m koh(aq). it requires 39.06 ml of the koh solution to produce a light pink indicator color. what is the molecular weight of the unknown acid?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Electrons are extremely important to what area of technology? a) anti-aging research b) household product development c) electronics d) drug discovery
Answers: 3


Chemistry, 22.06.2019 16:20
When water dissolves sugar, which process is not involved? o dissociation o hydration o surface area of the solute increases sa
Answers: 1

You know the right answer?
You are given a 1.00 g sample of an unknown tri-protic acid, which you dissolve in 50.0 ml of water...
Questions



Engineering, 22.02.2020 00:39

Social Studies, 22.02.2020 00:39



Computers and Technology, 22.02.2020 00:40







English, 22.02.2020 00:40

Mathematics, 22.02.2020 00:40




Mathematics, 22.02.2020 00:40