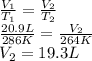An arctic weather balloon is filled with 20.9l of helium gas inside a prep shed. the temperature inside the shed is 13 degree c. the balloon is then taken outside, where the temperature is -9. degree c. calculate the new volume of the balloon. you may assume the pressure on the balloon stays constant at exactly 1atm. be sure your answer has the correct number of significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 3

Chemistry, 22.06.2019 12:30
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1


Chemistry, 23.06.2019 01:00
Heat energy, carbon dioxide, and water are released through which process? a. photosynthesis b. depolymerization c. digestion d. cellular respiration
Answers: 1
You know the right answer?
An arctic weather balloon is filled with 20.9l of helium gas inside a prep shed. the temperature ins...
Questions




History, 09.02.2021 20:50



Physics, 09.02.2021 20:50

Chemistry, 09.02.2021 20:50

Mathematics, 09.02.2021 20:50

Mathematics, 09.02.2021 20:50







Mathematics, 09.02.2021 20:50



English, 09.02.2021 20:50