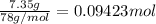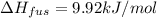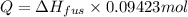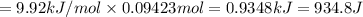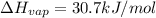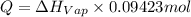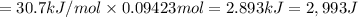Calculate the heat required to melt 7.35 g of benzene at its normal melting point. heat of fusion (benzene) = 9.92 kj/mol heat = kj
calculate the heat required to vaporize 7.35 g of benzene at its normal boiling point. heat of vaporization (benzene) = 30.7 kj/mol heat = kj

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Which of the following is a compound? a.carbon b.oxygen c.hydrogen d.water
Answers: 2

Chemistry, 21.06.2019 18:30
Asample of the male sex hormone testosterone, c19h28o2, contains 3.88×10^21 atoms of hydrogen.(a) how many atoms of carbon does it contain? (b) how many molecules of testosterone does it contain? (c) how many moles of testosterone does it contain? (d) what is the mass of this sample in grams?
Answers: 1

Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2

You know the right answer?
Calculate the heat required to melt 7.35 g of benzene at its normal melting point. heat of fusion (b...
Questions

English, 27.10.2020 22:50




Mathematics, 27.10.2020 22:50


History, 27.10.2020 22:50


Mathematics, 27.10.2020 22:50

Chemistry, 27.10.2020 22:50


Biology, 27.10.2020 22:50

Mathematics, 27.10.2020 22:50

English, 27.10.2020 22:50


Mathematics, 27.10.2020 22:50

Mathematics, 27.10.2020 22:50

Mathematics, 27.10.2020 22:50

History, 27.10.2020 22:50

Computers and Technology, 27.10.2020 22:50