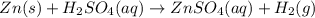Chemistry, 15.12.2019 05:31 Jaymp2706jj
How many moles of hydrogen gas will be produced if 5.00 moles of zinc reactions with an excess amount of sulfuric acid

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
When a spring is compressed, the energy changes from kinetic to potential. which best describes what is causing this change?
Answers: 3

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3

Chemistry, 22.06.2019 20:00
The volume of a single vanadium atom is 9.29×10-24 cm3. what is the volume of a vanadium atom in microliters?
Answers: 3

Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3
You know the right answer?
How many moles of hydrogen gas will be produced if 5.00 moles of zinc reactions with an excess amoun...
Questions

English, 04.10.2020 14:01


History, 04.10.2020 14:01


English, 04.10.2020 14:01




Social Studies, 04.10.2020 14:01


English, 04.10.2020 14:01

Mathematics, 04.10.2020 14:01



English, 04.10.2020 14:01

English, 04.10.2020 14:01

History, 04.10.2020 14:01