
Chemistry, 14.12.2019 07:31 andrejr0330jr
Nitrogen and oxygen can react to form various compounds. two experiments showed that one compound is formed when 3.62 g of nitrogen and 2.07 g of oxygen react completely, while another compound is formed when 1.82 g of nitrogen reacts completely with 4.13 g of oxygen. which of the following are most likely the molecular formulas for the nitrogen oxides obtained in these experiments? (1) no, n2o (2) no, no2 (3) n2o, n2o5 (4) no, n2o4 (5) n2o, n2o4

Answers: 2
Another question on Chemistry

Chemistry, 23.06.2019 02:20
Why dose heating increase the speed at which a solution dissolved in water
Answers: 1

Chemistry, 23.06.2019 03:30
Calculate the ph of a .10m nh4cl solution. the kb value for nh3 is 1.8×10^-5
Answers: 1

Chemistry, 23.06.2019 08:50
Reacting masses1 calcium carbonate breaks down on heating to produce calcium oxide and carbondioxide gas.caco3 + cao + co2a student heats 15 g of calcium carbonate strongly in a crucible.relative atomic masses (a): ca = 40, c = 12, o = 16.calculate the mass of calcium oxide produced by this reaction.(5 marks)
Answers: 3

Chemistry, 23.06.2019 11:20
Which of the following is a pure substance? airbloodcopperwood
Answers: 2
You know the right answer?
Nitrogen and oxygen can react to form various compounds. two experiments showed that one compound is...
Questions

Mathematics, 30.03.2020 03:16

Mathematics, 30.03.2020 03:17



History, 30.03.2020 03:26





Mathematics, 30.03.2020 03:26


Mathematics, 30.03.2020 03:26


Mathematics, 30.03.2020 03:26

Chemistry, 30.03.2020 03:27

English, 30.03.2020 03:27




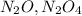
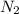 .
.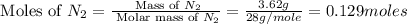
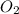 .
.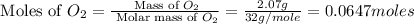
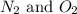 .
.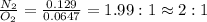
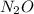 .
.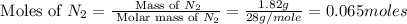
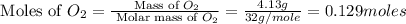
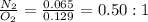
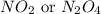 .
.

