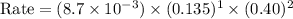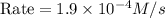Chemistry, 13.12.2019 23:31 albattatasraap5wymy
Consider the following reaction between mercury(ii) chloride and oxalate ion:
2hgcl2(aq)+c2o2−4(aq)→2cl−(aq)+2co2 (g)+hg2cl2(s)
the initial rate of this reaction was determined for several concentrations of hgcl2 and c2o2−4, and the following rate data were obtained for the rate of disappearance of c2o2−4:
experiment hgcl2(m) c2o2−4(m) rate (m/s)
1 0.164 0.15 3.2×10^−5
2 0.164 0.45 2.9×10^−4
3 0.082 0.45 1.4×10^−4
4 0.246 0.15 4.8×10^−5
what is the reaction rate when the concentration of hgcl2 is 0.135 m and that of c2o2−4 is 0.40 m , if the temperature is the same as that used to obtain the data shown?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

Chemistry, 22.06.2019 19:00
Which statement best describes what happens when molecular compounds melt
Answers: 1

Chemistry, 22.06.2019 22:00
What mass of glucose is produced when 54g of water react with carbon dioxide
Answers: 1
You know the right answer?
Consider the following reaction between mercury(ii) chloride and oxalate ion:
2hgcl2(aq)+c2o2...
2hgcl2(aq)+c2o2...
Questions

Mathematics, 03.03.2021 20:00

English, 03.03.2021 20:00

Physics, 03.03.2021 20:00

Mathematics, 03.03.2021 20:00

Social Studies, 03.03.2021 20:00




Chemistry, 03.03.2021 20:00

Social Studies, 03.03.2021 20:00


Mathematics, 03.03.2021 20:00

Social Studies, 03.03.2021 20:00

Social Studies, 03.03.2021 20:00




Health, 03.03.2021 20:00


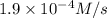
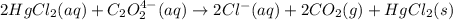
![\text{Rate}=k[HgCl_2]^a[C_2O_2^{4-}]^b](/tpl/images/0417/7719/2a902.png)
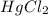
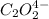
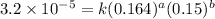 ....(1)
....(1)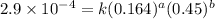 ....(2)
....(2)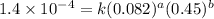 ....(3)
....(3)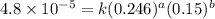 ....(4)
....(4)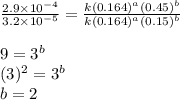
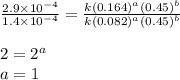
![\text{Rate}=k[HgCl_2]^1[C_2O_2^{4-}]^2](/tpl/images/0417/7719/2292e.png)
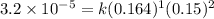
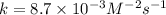
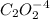 is 0.40 M.
is 0.40 M.