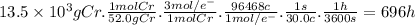Purification of chromium can be achieved by electrorefining chromium from an impure chromium anode onto a pure chromium cathode in an electrolytic cell. how many hours will it take to plate 13.5 kg of chromium onto the cathode if the current passed through the cell is held constant at 30.0 a ? assume the chromium in the electrolytic solution is present as cr 3 +.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Consider the point on the plot where 10.0 g of naoh have been added. what amount of naoh, in moles, has been added? 0.308 mol fecl3 initially present
Answers: 1

Chemistry, 23.06.2019 00:30
On the periodic table, elements are arranged by which of the following. a. mass numbers. b. increasing atomic number. c. alphabetical order. or d. density
Answers: 1

Chemistry, 23.06.2019 03:00
What does a complete balanced chemical equation include? a. exothermic coefficients b. endothermic coefficients c. valence electrons d. molar coefficients
Answers: 1

Chemistry, 23.06.2019 09:00
A2-kg bowling ball is 1 meter off the ground on a post when it falls. just before it reaches the ground,its traveling 4.4 m/s. assuming that there is no air resistant, which statement is true a. the initial potential energy is less then the final kinetic energy b. the mechanical energy is not conserved c. the mechanical energy is conserved d. the initial potential energy is greater than the final kinetic energy
Answers: 3
You know the right answer?
Purification of chromium can be achieved by electrorefining chromium from an impure chromium anode o...
Questions



History, 21.09.2019 18:50


Mathematics, 21.09.2019 18:50

Mathematics, 21.09.2019 18:50



Social Studies, 21.09.2019 18:50


Chemistry, 21.09.2019 18:50

History, 21.09.2019 18:50

History, 21.09.2019 18:50





History, 21.09.2019 18:50

History, 21.09.2019 18:50

Mathematics, 21.09.2019 18:50