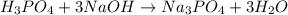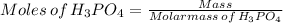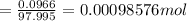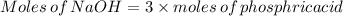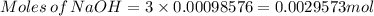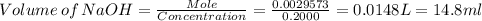Achemistry student weighs out of phosphoric acid , a triprotic acid, into a volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with solution. calculate the volume of solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Rutherford's experiment indicated that matter was not as uniform as it appears what part of his experimental results implied this idea
Answers: 1

Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 14:30
Aroom with dimensions 7.00m×8.00m×2.50m is to be filled with pure oxygen at 22.0∘c and 1.00 atm. the molar mass of oxygen is 32.0 g/mol. how many moles noxygen of oxygen are required to fill the room? what is the mass moxygen of this oxygen?
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3
You know the right answer?
Achemistry student weighs out of phosphoric acid , a triprotic acid, into a volumetric flask and dil...
Questions

Biology, 27.07.2019 15:30


Business, 27.07.2019 15:30


History, 27.07.2019 15:30



Physics, 27.07.2019 15:30


Mathematics, 27.07.2019 15:30




History, 27.07.2019 15:30

Social Studies, 27.07.2019 15:30

Biology, 27.07.2019 15:30


Social Studies, 27.07.2019 15:30

Social Studies, 27.07.2019 15:30