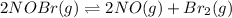Chemistry, 12.12.2019 04:31 dhurtado1195
For the following reaction, the equilibrium constant kc is 2.0 at a certain temperature. the reaction is endothermic. what do you expect to happen to the concentration of no if the temperature is doubled? 2nobr( g) ⇌ 2no( g) + br 2( g) the change in concentration of [no] will depend on the size of the vessel. the concentration of no will increase. the concentration of no will decrease. a catalyst will be needed to make a change in concentration. there will be no change in [no].

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:30
You are to give ampicillin with a recommended dose of 25mg/kg to a child with a mass of 29kg. if stock on hand is 250mg/capsule how many capsules should be given?
Answers: 1


Chemistry, 22.06.2019 06:00
Calculate the mass of silver needed to react with chlorine to produce 126g if silver chloride?
Answers: 3

Chemistry, 22.06.2019 07:30
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3
You know the right answer?
For the following reaction, the equilibrium constant kc is 2.0 at a certain temperature. the reactio...
Questions

Mathematics, 09.06.2020 03:57

Mathematics, 09.06.2020 03:57

English, 09.06.2020 03:57

Health, 09.06.2020 03:57

Physics, 09.06.2020 03:57

Mathematics, 09.06.2020 03:57




English, 09.06.2020 03:57






Mathematics, 09.06.2020 03:57


Chemistry, 09.06.2020 03:57

Mathematics, 09.06.2020 03:57

History, 09.06.2020 03:57