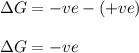Chemistry, 12.12.2019 04:31 keasiabradley
During an exothermic chemical reaction, a solid is consumed and a gas produced. a. yes b. no c. can't decide with information given.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1

Chemistry, 22.06.2019 12:30
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 22.06.2019 22:40
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1
You know the right answer?
During an exothermic chemical reaction, a solid is consumed and a gas produced. a. yes b. no c. can'...
Questions


Computers and Technology, 19.11.2020 19:00

Mathematics, 19.11.2020 19:00

Mathematics, 19.11.2020 19:00



Mathematics, 19.11.2020 19:00







Biology, 19.11.2020 19:00


English, 19.11.2020 19:00


Biology, 19.11.2020 19:00

Mathematics, 19.11.2020 19:00

Mathematics, 19.11.2020 19:00

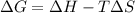
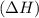 of the reaction comes out to be negative for this kind of reaction.
of the reaction comes out to be negative for this kind of reaction.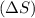 . Randomness of gaseous particles is more than that of liquid which is further more than that of solids.
. Randomness of gaseous particles is more than that of liquid which is further more than that of solids.