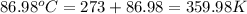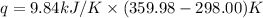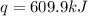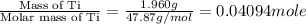Chemistry, 11.12.2019 23:31 cheyennemitchel2680
When 1.960 g of titanium is combusted in a bomb calorimeter, the temperature of the calorimeter increases from 25.00 °c to 86.98 °c. in a separate experiment, the heat capacity of the calorimeter is measured to be 9.84 kj/k. the heat of reaction for the combustion of a mole of ti in this calorimeter is kj/mol.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 19:00
How many moles of cu are needed to react with 5.8 moles of agno3? cu + 2 agno3 → cu(no3)2 + 2 ag
Answers: 3

Chemistry, 22.06.2019 19:10
Astudent completes a titration by adding 12.0 milliliters of naoh(aq) of unknown concentration to 16.0 milliliters of 0.15 m hcl(aq). what is the molar concentration of the naoh(aq)? 1)5.0 m 2)0.20 m 3)0.11 m 4)1.1 m
Answers: 1

Chemistry, 22.06.2019 22:00
Ill give u brainliest pls how is mass of carbon conserved during cellular respiration
Answers: 1
You know the right answer?
When 1.960 g of titanium is combusted in a bomb calorimeter, the temperature of the calorimeter incr...
Questions






Social Studies, 12.03.2022 05:00




Mathematics, 12.03.2022 05:00



History, 12.03.2022 05:00




Mathematics, 12.03.2022 05:00



Biology, 12.03.2022 05:10

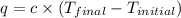
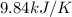
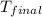 = final temperature =
= final temperature = 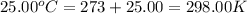
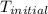 = initial temperature =
= initial temperature =