
Chemistry, 11.12.2019 04:31 naynay4evr
Click in the answer box to activate the palette. use the half-reaction method to balance the following equation in acidic solution. it is not necessary to include any phases of matter for any species. cn− + mno4− → cno− + mno2

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:30
If i make a solution by adding 83grams of sodium hydroxide to 750ml i’d water what is the molarity of sodium hydroxide
Answers: 1

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3

Chemistry, 22.06.2019 02:30
Asa choose the correct set of reaction coefficients to properly balance the following chemical equation according to the law of conservation of mass: __s8 + __o2 ==> __so2 1, 1, 8 1, 8, 1 1, 8, 8 8, 1, 1
Answers: 1

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1
You know the right answer?
Click in the answer box to activate the palette. use the half-reaction method to balance the followi...
Questions



English, 02.09.2019 08:30

Mathematics, 02.09.2019 08:30

Social Studies, 02.09.2019 08:30





Computers and Technology, 02.09.2019 08:30

Social Studies, 02.09.2019 08:30

Social Studies, 02.09.2019 08:30


Health, 02.09.2019 08:30


Biology, 02.09.2019 08:30

History, 02.09.2019 08:30

Social Studies, 02.09.2019 08:30


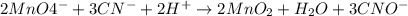
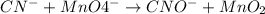
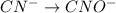 ( oxidation)
( oxidation)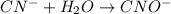
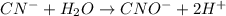
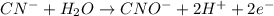 ..[1]
..[1]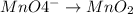 (reduction)
(reduction)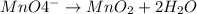
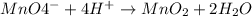 ..[2]
..[2]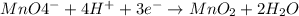 ..[2]
..[2]

