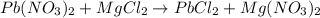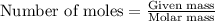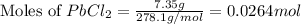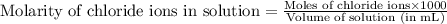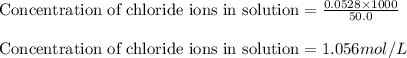Chemistry, 11.12.2019 04:31 QueenNerdy889
A12.00g sample of addition of an excess of lead (ii) nitrate to a 50.0ml solution of magnesium chloride caused a formation of 7.35g of lead (ii) chloride precipitate. what was the molar concentration of chloride ions in the solution (in mol/l)?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 09:10
Select the correct answer from each drop-down menu.describe what happens to a carbon-11 atom when it undergoes positron emission.the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3

Chemistry, 22.06.2019 12:30
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1

You know the right answer?
A12.00g sample of addition of an excess of lead (ii) nitrate to a 50.0ml solution of magnesium chlor...
Questions





Mathematics, 12.02.2021 17:50

Mathematics, 12.02.2021 17:50

Mathematics, 12.02.2021 17:50

Chemistry, 12.02.2021 17:50



Mathematics, 12.02.2021 17:50

Chemistry, 12.02.2021 17:50

English, 12.02.2021 17:50



Mathematics, 12.02.2021 17:50



English, 12.02.2021 17:50

English, 12.02.2021 17:50