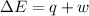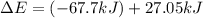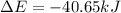Chemistry, 10.12.2019 06:31 wendelkristen
Consider a balloon filled with helium at the following conditions. 313 g he 1.00 atm 1910. l molar heat capacity = 20.8 j/degree c middot mol the temperature of this balloon is decreased by 41.6 degree c as the volume decreases to 1643 l with the pressure remaining constant. determine q, w, and delta e (in kj) for the compression of the balloon.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 16:00
Which of the following is the correct definition of chemical energy? a. energy an object has because of its motion or position b. energy resulting from the flow of charged particles, such as electrons or ions c. energy produced from the splitting of atoms d. energy stored in chemical bonds of molecules
Answers: 1

Chemistry, 23.06.2019 02:00
As light moves from one material into the next, which of the following affects how much the light waves will refract, or bend? angle at which the ray strikes the medium color of the material density of the material temperature of the light wave
Answers: 2

Chemistry, 23.06.2019 02:00
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1
You know the right answer?
Consider a balloon filled with helium at the following conditions. 313 g he 1.00 atm 1910. l molar h...
Questions







Biology, 05.01.2022 19:40




Mathematics, 05.01.2022 19:40


SAT, 05.01.2022 19:40

Mathematics, 05.01.2022 19:40



Mathematics, 05.01.2022 19:50

Mathematics, 05.01.2022 19:50


Mathematics, 05.01.2022 19:50

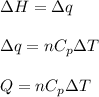
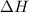 = change in enthalpy energy
= change in enthalpy energy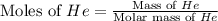
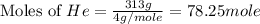
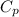 = heat capacity at constant pressure =
= heat capacity at constant pressure = 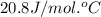
 = change in temperature =
= change in temperature = 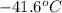
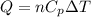
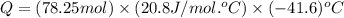
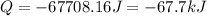
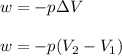
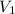 = initial volume = 1910 L
= initial volume = 1910 L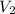 = final volume = 1643 L
= final volume = 1643 L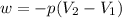
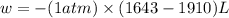
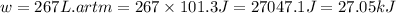
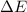 of the gas.
of the gas.