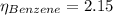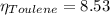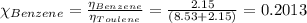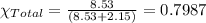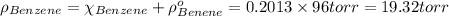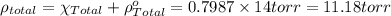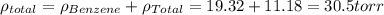In a mixture of volatile substances, the vapor pressure of the solution depends on the vapor pressure of both substances (solute and solvent). each component can evaporate, but its vapor pressure is lowered by the presence of the other substance following rault's law. the vapor pressure of the solvent is calculated using psolvent = xsolvent*posolvent and the vapor pressure of the solute is calculated using psolute = xsolute*posolute. the total vapor pressure can be calculated by dalton's law of partial pressures, ptotal = psolvent + psolute. at 25oc, the vapor pressure of pure benzene (c6h6, 78.11 g/mol) is 96 torr. at the same temperature, the vapor pressure of pure toluene (c7h8, 92.14 g/mol) is 14 torr. consider a solution containing 3.71 mol of benzene and 5.52 mol of toluene. calculate the vapor pressure above the solution. enter your answer in units of torr to three significant figures.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1

Chemistry, 22.06.2019 23:00
What is the oxidation state of an individual bromine atom in nabro3?
Answers: 2

Chemistry, 23.06.2019 01:00
Which of the following is in the lanthanide family? a) uranium b) promethium c) silver d) gold
Answers: 2

Chemistry, 23.06.2019 05:00
He nucleus contains the cells genetic material in the form of dna. dna is organized into our chromosomes, which are made up of thousands of that determine our traits.
Answers: 1
You know the right answer?
In a mixture of volatile substances, the vapor pressure of the solution depends on the vapor pressur...
Questions





Mathematics, 16.06.2021 18:20



Arts, 16.06.2021 18:20


Mathematics, 16.06.2021 18:20

Mathematics, 16.06.2021 18:20




Social Studies, 16.06.2021 18:20

Engineering, 16.06.2021 18:20



Law, 16.06.2021 18:20