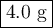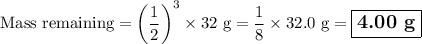Chemistry, 10.12.2019 02:31 angellll4455
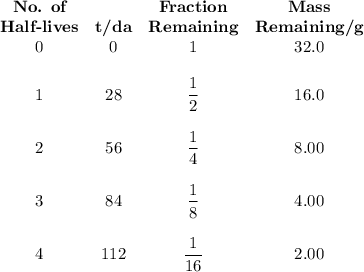
9.) the isotope chromium-51 has a half-life of 28 days. if the original sample had a 51cr mass of 32.0 grams, what mass of 51cr would remain after 84 days

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 19:30
Which one of the following substances would be the most soluble in ccl4? na2so4 h2o ch3ch2ch2ch2oh c4h10 hi
Answers: 1


Chemistry, 23.06.2019 05:30
The image compares the arrangement of electrons in two different neutral atoms. a figure labeled atom q has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has six black spheres. to the left of this figure is another figure labeled atom p. atom p has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has seven black spheres. which of the following best explains the position of the two atoms in the periodic table? atom p has an estimated zeff of 7 and is therefore to the left of atom q, which has a zeff of 6. atom p has an estimated zeff of 7 and is therefore to the right of atom q, which has a zeff of 6. atom p has an estimated zeff of 5 and is therefore below atom q, which has a zeff of 4. atom p has an estimated zeff of 5 and is therefore above atom q, which has a zeff of 4.
Answers: 3

You know the right answer?
9.) the isotope chromium-51 has a half-life of 28 days. if the original sample had a 51cr mass of 32...
Questions


Chemistry, 17.07.2019 08:30

Mathematics, 17.07.2019 08:30

Computers and Technology, 17.07.2019 08:30

Mathematics, 17.07.2019 08:30




Mathematics, 17.07.2019 08:30


Mathematics, 17.07.2019 08:30

Chemistry, 17.07.2019 08:30




Mathematics, 17.07.2019 08:30




English, 17.07.2019 08:30