
Chemistry, 07.12.2019 03:31 jc95704816
Balance the chemical equation given below, and determine the number of milliliters of 0.0300m phosphoric acid required to neutralize 25.00ml of 0.0150m calcium hydroxide.
__ca(oh)2(aq) + __h3po4(aq) --> __ca3(po4)2(s) + __h2o(l)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which part of a feedback mechanism is able to monitor the conditions outside of cells and usually uses nerve cells to relay this information to an intergrating center
Answers: 2

Chemistry, 22.06.2019 06:00
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 07:00
Which atom or ion is the largest? a. k b. k+ c. ca d. ca2+ e. li
Answers: 1

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2
You know the right answer?
Balance the chemical equation given below, and determine the number of milliliters of 0.0300m phosph...
Questions

Computers and Technology, 25.02.2020 00:05





Mathematics, 25.02.2020 00:05





Mathematics, 25.02.2020 00:05







Geography, 25.02.2020 00:05


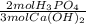 = 2,50x10⁻⁴ moles of H₃PO₄.
= 2,50x10⁻⁴ moles of H₃PO₄. 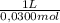 = 8,33x10⁻³L ≡ 8,33mL of 0,0300M phosphoric acid
= 8,33x10⁻³L ≡ 8,33mL of 0,0300M phosphoric acid

