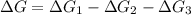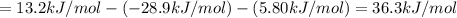Consider these hypothetical chemicalreactions:
{\rm a \rightleftharpoons b}, \quad\delta g =...

Chemistry, 07.12.2019 02:31 queenkimm26
Consider these hypothetical chemicalreactions:
{\rm a \rightleftharpoons b}, \quad\delta g = 13.2 kj/mol
{\rm b \rightleftharpoons c}, \quad\delta g = -28.9 kj/mol
{\rm c \rightleftharpoons d},\quad \delta g = 5.80 kj/mol
what is the free energy, delta g, for the overall reaction, \rm a \rightleftharpoons d ?
express your answer numerically inkilojoules per mole.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 07:50
Which of the following electromagnetic waves can create ions?
Answers: 2

Chemistry, 22.06.2019 09:00
The nuclear fission process releases neutrons and question 27 options: alpha particles electrons energy beta particles
Answers: 1

Chemistry, 22.06.2019 11:00
The number to the right of an element's symbol (ex. c-12) identifies the of an isotope.
Answers: 1
You know the right answer?
Questions






Mathematics, 28.07.2021 14:00

Biology, 28.07.2021 14:00


Physics, 28.07.2021 14:00

Mathematics, 28.07.2021 14:00

Mathematics, 28.07.2021 14:00

Mathematics, 28.07.2021 14:00



English, 28.07.2021 14:00



Mathematics, 28.07.2021 14:00

Business, 28.07.2021 14:00

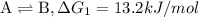 ...[1]
...[1]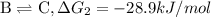 ...[2]
...[2]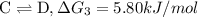 ...[3]
...[3]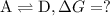 ...[4]
...[4]