Based on your data, what is the likely identity (complete chemical formula) of the unknown sulfate? explain (include calculations as appropriate).
mass (g) of unknown sulfate = 0.53g
mass g of crucible= 28.63g
mass g of crucible + content= 29.44 g
mass of barium sulfate = .81g

Answers: 1
Another question on Chemistry


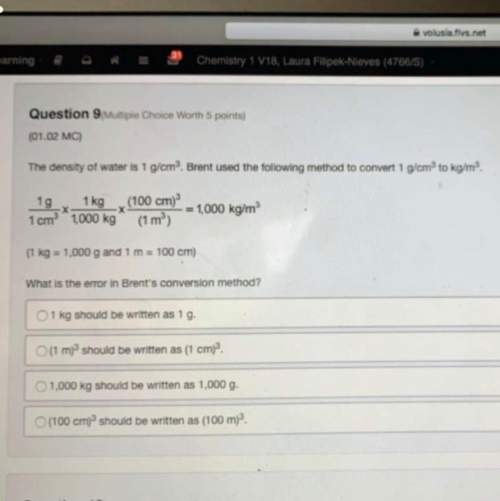
Chemistry, 22.06.2019 00:30
13. calculate the initial concentration (before precipitation) of carbonate ions after the addition of each 0.05 ml of solution b to the 1.00 l beaker of solution a. divide the work among group members and write the answers in the table in model 3. assume the volume change as solution b is added is negligible. 14. notice the initial concentrations of zn2+ - and cu2+ in the table in model 3. a. explain how these were obtained from the data in model 2. b. as solution b is added and precipitates form, do these initial concentrations change? 15. use the data in model 2 to indicate the presence of precipitate (either znco3 or cuco3) after each 0.05 ml addition of solution b in model 3. 16. use the initial concentrations of carbonate ions and zinc ions to calculate the reaction quotient, qsp for the zinc carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3. 17. use the initial concentrations of carbonate ion and copper(ii) ions to calculate the qsp for the copper(ii) carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3.
Answers: 3

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

Chemistry, 22.06.2019 19:00
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2
You know the right answer?
Based on your data, what is the likely identity (complete chemical formula) of the unknown sulfate?...
Questions

Chemistry, 09.07.2019 20:30

History, 09.07.2019 20:30



Social Studies, 09.07.2019 20:30




English, 09.07.2019 20:30

English, 09.07.2019 20:30

Mathematics, 09.07.2019 20:30





Mathematics, 09.07.2019 20:30



Mathematics, 09.07.2019 20:30

English, 09.07.2019 20:30