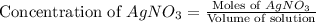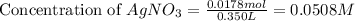Chemistry, 06.12.2019 19:31 orlando19882000
Suppose a 2.95 g of potassium iodide is dissolved in 350. ml of a 62.0 m m aqueous solution of silver nitrate. calculate the final molarity of iodide anion in the solution. you can assume the volume of the solution doesn't change when the potassium iodide is dissolved in it. be sure your answer has the correct number of significant digits

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 01:30
What is the value of keq for the reaction expressed in scientific notation
Answers: 1


Chemistry, 22.06.2019 21:30
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1
You know the right answer?
Suppose a 2.95 g of potassium iodide is dissolved in 350. ml of a 62.0 m m aqueous solution of silve...
Questions

Mathematics, 04.11.2020 01:00

Mathematics, 04.11.2020 01:00

English, 04.11.2020 01:00

Mathematics, 04.11.2020 01:00

Social Studies, 04.11.2020 01:00


History, 04.11.2020 01:00

Biology, 04.11.2020 01:00

Mathematics, 04.11.2020 01:00



Biology, 04.11.2020 01:00



History, 04.11.2020 01:00

Biology, 04.11.2020 01:00

Mathematics, 04.11.2020 01:00

English, 04.11.2020 01:00


Social Studies, 04.11.2020 01:00

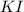 and
and 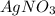 .
.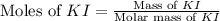
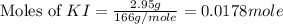
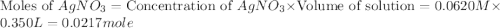
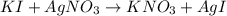
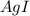
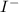 anion = Moles of
anion = Moles of 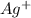 cation = 0.0178 moles
cation = 0.0178 moles