
Give the chemical symbol for the element with the ground‑state electron configuration [ ar ] 4 s 2 3 d 1 . symbol: determine the quantum numbers n and ℓ and select all possible values for m ℓ for each subshell of the element. 4 s n = 4 s ℓ = the possible values of m ℓ for the 4 s subshell are 0 − 1 , 0 , + 1 − 2 , − 1 , 0 , + 1 , + 2 − 3 , − 2 , − 1 , 0 , + 1 , + 2 , + 3 3 d n = 3 d ℓ = the possible values of m ℓ for the 3 d subshell are − 1 , 0 , + 1 − 2 , − 1 , 0 , + 1 , + 2 − 3 , − 2 , − 1 , 0 , + 1 , + 2 , + 3 0

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:00
Nickel crystallizes in the face-centered cubic (fcc) lattice. the density of the metal is 8902 kg/m3. calculate the radius of a nickel atom.
Answers: 1

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 21.06.2019 23:30
For the following dehydrohalogenation (e2) reaction, draw the zaitsev product(s) resulting from elimination involving c3–c4 (i.e., the carbon atoms depicted with stereobonds). show the product stereochemistry clearly. if there is more than one organic product, both products may be drawn in the same box. ignore elimination involving c3 or c4 and any carbon atom other than c4 or c3.
Answers: 3

You know the right answer?
Give the chemical symbol for the element with the ground‑state electron configuration [ ar ] 4 s 2 3...
Questions









Computers and Technology, 11.03.2020 05:37






Computers and Technology, 11.03.2020 05:38

Mathematics, 11.03.2020 05:38




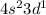 .
.

