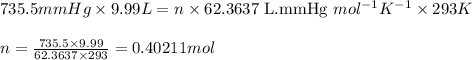Oxygen gas can be prepared by heating potassium chlorate according to the following equation: 2kclo3(s)arrow. gif2kcl(s) + 3o2(g)the product gas, o2, is collected over water at a temperature of 20 °c and a pressure of 748 mm hg. if the wet o2 gas formed occupies a volume of 9.49 l, the number of moles of kclo3 reacted was ? mol. the vapor pressure of water is 17.5 mm hg at 20 °c. oxygen gas can be prepared by heating potassium chlorate according to the following equation: 2kclo3(s)arrow. gif2kcl(s) + 3o2(g)the product gas, o2, is collected over water at a temperature of 20 °c and a pressure of 753 mm hg. if the wet o2 gas formed occupies a volume of 9.99 l, the number of grams of o2 formed is ? g. the vapor pressure of water is17.5 mm hg at 20 °c.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 13:30
One of the reactions in a blast furnace used to reduce iron is shown above. how many grams of fe2o3 are required to produce 15.5 g of fe if the reaction occurs in the presence of excess co? a.11.1 g b.22.1 g c.30.0 g d.44.2 g
Answers: 2

Chemistry, 21.06.2019 21:10
Identify one disadvantage to each of the following models of electron configuration: dot structures arrow and line diagrams written electron configurations type in your answer below.
Answers: 1

Chemistry, 22.06.2019 03:50
Which of the following statements about acidic water is true? a. acid has no effect on the h,o molecules. b. the solution contains a larger number of oh ions than h,o ions. c. the solution contains a larger number of h,o ions than qh ions. d. the solution contains an equal number of h,o ions and oh ions. none of the above e.
Answers: 1

You know the right answer?
Oxygen gas can be prepared by heating potassium chlorate according to the following equation: 2kclo3...
Questions

Mathematics, 03.09.2020 16:01

Mathematics, 03.09.2020 16:01

Mathematics, 03.09.2020 16:01


English, 03.09.2020 16:01



Business, 03.09.2020 16:01

Computers and Technology, 03.09.2020 16:01

Spanish, 03.09.2020 16:01



English, 03.09.2020 16:01





Mathematics, 03.09.2020 16:01


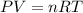
![20^oC=[20+273]K=293K](/tpl/images/0406/0694/3b5d4.png)
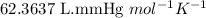
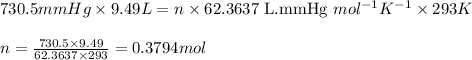
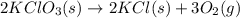
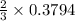 moles of potassium chlorate undergoes reaction.
moles of potassium chlorate undergoes reaction.