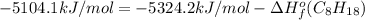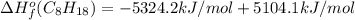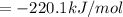Chemistry, 06.12.2019 05:31 breannamiller0822
Or a particular isomer of c 8 h 18 , the combustion reaction produces 5104.1 kj of heat per mole of c 8 h 18 ( g ) consumed, under standard conditions. c 8 h 18 ( g ) + 25 2 o 2 ( g ) ⟶ 8 co 2 ( g ) + 9 h 2 o ( g ) δ h ∘ rxn = − 5104.1 kj / mol what is the standard enthalpy of formation of this isomer of c 8 h 18 ( g ) ?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:20
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 12:40
Consider the directing effects of the substituents on salicylamide and predict the possible structures of the iodination products. which do you think will be the major product?
Answers: 1

Chemistry, 22.06.2019 22:00
The diagrams to the right show the distribution and arrangement of gas particles in two different containers. according to kinetic-molecular theory, which of the following statements is true? check all that apply. if the temperatures of both containers are equal, container a has greater pressure than container b. if the volume of container a decreased, its pressure would decrease. if the pressure in both containers is equal, container a has a lower temperature than container b.
Answers: 2

Chemistry, 22.06.2019 22:30
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1
You know the right answer?
Or a particular isomer of c 8 h 18 , the combustion reaction produces 5104.1 kj of heat per mole of...
Questions

Mathematics, 29.10.2020 23:20

English, 29.10.2020 23:20

Mathematics, 29.10.2020 23:20




Mathematics, 29.10.2020 23:20

Mathematics, 29.10.2020 23:20





Arts, 29.10.2020 23:20


Social Studies, 29.10.2020 23:20


Social Studies, 29.10.2020 23:20


Geography, 29.10.2020 23:20

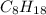 is -220.1 kJ/mol.
is -220.1 kJ/mol.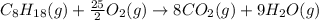
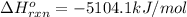
![\Delta H^{o}_{rxn}=[8\Delta H^{o}_{f}(CO_{2}) +9\Delta H^{o}_{f}(H_{2}O)]-[\Delta H^{o}_{f}(C_{8}H_{18})+ \frac{25}{2}\Delta H^{o}_{f}(O_{2})]](/tpl/images/0405/9197/1d3a2.png)
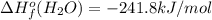
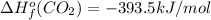
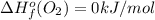
![-5104.1kJ/mol=[8(-393.5)+9(-241.8)kJ/mol]-[\Delta H^{o}_{f}(C_{8}H_{18})+ \frac{25}{2}(0)kJ/mol]](/tpl/images/0405/9197/edc02.png)