
Chemistry, 06.12.2019 05:31 DisneyyKayy
Calculate δh∘ in kilojoules for the reaction of acetylene (c2h2) (δh∘f=227.4kj/mol) with o2 to yield carbon dioxide (co2) (δh∘f=−393.5 kj/mol) and h2o(g) (δh∘f=−241.8kj/mol), a reaction which is supplied by the industrial gases industry for oxyacetylene gas welding and cutting due to the high temperature of the flame.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Check the correct box to describe the periodic trends in electronegativity. electronegativity across a period: decreases. increases. electronegativity down a group: decreases. increases.
Answers: 2

Chemistry, 22.06.2019 09:40
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2

Chemistry, 22.06.2019 12:30
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3

Chemistry, 22.06.2019 14:10
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2
You know the right answer?
Calculate δh∘ in kilojoules for the reaction of acetylene (c2h2) (δh∘f=227.4kj/mol) with o2 to yield...
Questions


Mathematics, 16.11.2020 02:10


Mathematics, 16.11.2020 02:10

Mathematics, 16.11.2020 02:10


Mathematics, 16.11.2020 02:10


Mathematics, 16.11.2020 02:10


Mathematics, 16.11.2020 02:20

Mathematics, 16.11.2020 02:20

History, 16.11.2020 02:20


Chemistry, 16.11.2020 02:20



Mathematics, 16.11.2020 02:20

English, 16.11.2020 02:20

Physics, 16.11.2020 02:20

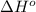 for the reaction is, -2512.4 kJ
for the reaction is, -2512.4 kJ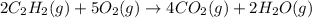
![\Delta H^o_{rxn}=[(3\times \Delta H^o_f_{(CO_2(g))})+(4\times \Delta H^o_f_{(H_2O(g))})]-[(1\times \Delta H^o_f_{(C_2H_2(g))})+(5\times \Delta H^o_f_{(O_2(g))})]](/tpl/images/0405/9362/62f19.png)
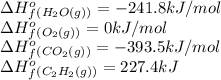
![\Delta H^o_{rxn}=[(4\times (-393.5))+(2\times (-241.8))]-[(2\times (227.4)+(5\times (0))]\\\\\Delta H^o_{rxn}=-2512.4kJ](/tpl/images/0405/9362/361f8.png)


