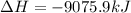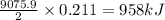Chemistry, 06.12.2019 05:31 lindseylewis313
B5h9(l) is a colorless liquid that will explode when exposed to oxygen. how much heat is released when 0.211 mol of b5h9 reacts with excess oxygen where the products are b2h3(s) and h2o(l). the standard enthalpy of formation of b5h9(l) is 73.2 kj/mol, the standard enthalpy of formation of b2h3(s) is -1272 kj/mol and that of h2o(l) is -285.4 kj/mol. express your answer in kj.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2

Chemistry, 22.06.2019 09:40
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1


You know the right answer?
B5h9(l) is a colorless liquid that will explode when exposed to oxygen. how much heat is released wh...
Questions

Geography, 05.11.2020 17:30



English, 05.11.2020 17:30




Biology, 05.11.2020 17:30



English, 05.11.2020 17:40


Mathematics, 05.11.2020 17:40

Mathematics, 05.11.2020 17:40

Mathematics, 05.11.2020 17:40

French, 05.11.2020 17:40

Mathematics, 05.11.2020 17:40

Spanish, 05.11.2020 17:40


Mathematics, 05.11.2020 17:40

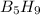 reacts with excess oxygen.
reacts with excess oxygen.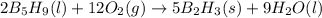
![\Delta H=\sum [n\times \Delta H_f(product)]-\sum [n\times \Delta H_f(reactant)]](/tpl/images/0405/9477/76c37.png)
![\Delta H=[(n_{B_2H_3}\times \Delta H_{B_2H_3})+(n_{H_2O}\times \Delta H_{H_2O})]-[(n_{O_2}\times \Delta H_{O_2})+(n_{B_5H_9}\times \Delta H_{B_5H_9})]](/tpl/images/0405/9477/bc69c.png)
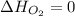 (as heat of formation of substances in their standard state is zero
(as heat of formation of substances in their standard state is zero![\Delta H=[(5\times -1272)+(9\times -285.5]-[(12\times 0)+(2\times 73.2)]](/tpl/images/0405/9477/56397.png)