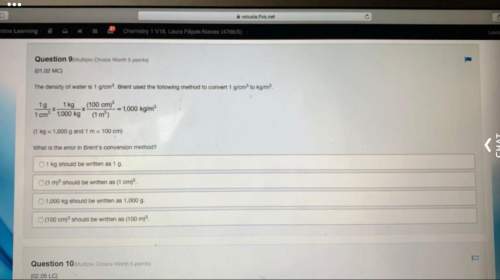
Amixture of 0.10 mol of no, 0.050 mol of h2, and 0.10 mol of h2o is placed in a 1.0-l vessel at 300 k. the following equilibrium is established: 2no(g)+2h2(g)⥫⥬==n2(g)+2h2o(g) at equilibrium [no]=0.062m. calculate the equilibrium concentrations of h2, n2, and h2o.
calculate the equilibrium concentration of n2

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 10:30
How do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 2

Chemistry, 22.06.2019 11:00
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

You know the right answer?
Amixture of 0.10 mol of no, 0.050 mol of h2, and 0.10 mol of h2o is placed in a 1.0-l vessel at 300...
Questions


Mathematics, 20.01.2021 20:20











English, 20.01.2021 20:20

Mathematics, 20.01.2021 20:20



Mathematics, 20.01.2021 20:20

Business, 20.01.2021 20:20

Mathematics, 20.01.2021 20:20