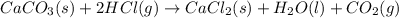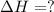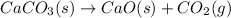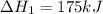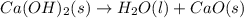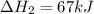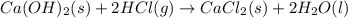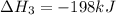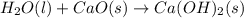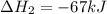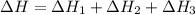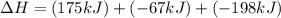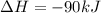Consider the following chemical equations to answer the question that follows. caco3(s)ca(oh)2(s)ca(oh)2(s)+2hcl(g )→cao(s)+co2(g)→h2o(l)+cao(s)→cacl2 (s)+2h2o(l)δhδhδh=175kj=67kj=−198kj using the information above, determine the change in enthalpy for the following chemical reaction. caco3(s)+2hcl(g)⟶cacl2(s)+h2o(l)+co 2(g)

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 07:20
2pos suppose an object in free fall is dropped from a building. its starting velocity is 0 m/s. ignoring the effects of air resistance, what is the speed (in m/s) of the object after falling 3 seconds? give your answer as a positive decimal without units. answer here
Answers: 1

Chemistry, 23.06.2019 04:10
What does the field of thermodynamics relate to a-changes in nuclear reactions b- changes in energy in systems c changes in molecular structure d changes in atomic properties
Answers: 1

Chemistry, 23.06.2019 06:00
Jenny wants to test the electrical conductivity of two substances dissolved in water. she is preparing the containers for the experiment. which factor is most important for her to control?
Answers: 1
You know the right answer?
Consider the following chemical equations to answer the question that follows. caco3(s)ca(oh)2(s)ca(...
Questions



Mathematics, 18.02.2021 01:00



Chemistry, 18.02.2021 01:00


History, 18.02.2021 01:00


Mathematics, 18.02.2021 01:00



Mathematics, 18.02.2021 01:00

Mathematics, 18.02.2021 01:00




Computers and Technology, 18.02.2021 01:00

History, 18.02.2021 01:00

History, 18.02.2021 01:00